Japan Plasma-Derived Therapeutics Market Growth Projection US$3.53Bn by 2033, Growth in IVIg & Key Pharma Companies 2026
Japan Plasma-Derived Therapeutics Market Insights, Hospital Demand & Biopharma Innovations (2026-2033)
Japan Plasma-Derived Therapeutics Market Growth, Plasma Collection Trends & Strategic Investments”
OSAKA, TOKYO, JAPAN, March 6, 2026 /EINPresswire.com/ -- Market Size and Growth (2026-2033)— DataM Intelligence 4Market Research LLP
Japan’s plasma-derived therapeutics market is accelerating with aging demographics, robust hospital demand, regulatory tailwinds, and high-value global partnerships - a prime growth opportunity for investors and industry leaders alike.
Market Overview
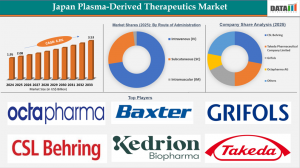
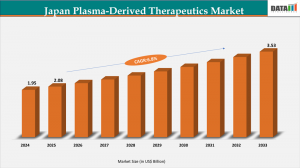
The Japan Plasma-Derived Therapeutics Market Size 2026 expanded from US$1.95 billion in 2024 to US$2.08 billion in 2025 and is forecast to reach US$3.53 billion by 2033, advancing at a CAGR of 6.8% over the forecast period (2026–2033). Rising prevalence of chronic diseases, increasing hospital-based infusions, and domestic plasma collection infrastructure are major drivers.
Get Sample Report - Preview key market trends and strategic insights in minutes:- https://www.datamintelligence.com/download-sample/japan-plasma-derived-therapeutics-market
Key products include:
1. Immunoglobulins (IVIg)
2. Albumin
3. Coagulation factors
The market is further strengthened by Japan’s universal healthcare system, which ensures broad patient access, and a regulated domestic plasma collection network maintaining high safety standards.
Market Drivers & Opportunities
Aging Population & Rising Clinical Demand - Chronic conditions in older adults fuel demand for plasma-derived therapies.
Technological Innovations in Fractionation - AI-assisted purification and viral safety technologies enhance product reliability.
Regulatory & Policy Support - Streamlined approval pathways and reimbursement schemes encourage adoption.
Strategic Collaborations & Supply Chain Expansion - Partnerships with global manufacturers reduce import dependency.
FDA Approvals of Plasma-Derived Therapeutics
1. 2025–2026 U.S. Updates: The FDA approved next-generation immunoglobulin therapies for rare autoimmune diseases, shortening review timelines.
2. Enhanced safety testing requirements for plasma products were introduced, ensuring higher patient protection while expediting market entry.
3. Impact: Streamlined approvals have increased U.S. market access and global demand, influencing Japan’s regulatory alignment.
Customized Research - Get tailored intelligence for strategic decision-making:- https://www.datamintelligence.com/customize/japan-plasma-derived-therapeutics-market
Market Segmentation 2026
The Intravenous (IV) route emerged as the leading segment in Japan’s Plasma-Derived Therapeutics Market in 2025, contributing 74% of total revenue.
By Product Type: Immunoglobulins, Coagulation Factors, Albumin, Protease Inhibitors, Others
By Application: Multiple Sclerosis, Neuropathies, Primary Immunodeficiency Disorders, Hemophilia, Autoimmune Diseases, Critical Care & Trauma, Others
By End User: Hospitals, Specialty Clinics, Homecare Settings, Ambulatory Surgical Centers, Others
By Source of Plasma: Whole Blood Plasma, Apheresis Plasma, Recovered Plasma
By Route of Administration: Intravenous, Subcutaneous, Intramuscular
Regulatory Affairs of Plasma-Derived Therapeutics
1. Japan implemented faster review processes for therapies addressing rare or critical conditions.
2. Regulatory harmonization with global standards (EMA & FDA) allows smoother cross-border collaborations.
3. Companies must comply with stringent GMP, GTP, and safety protocols, ensuring high-quality product outputs.
4. Implication: Regulatory clarity encourages innovation and reduces time-to-market for new plasma therapies.
Government Policies of Plasma-Derived Therapeutics
. Japan’s Ministry of Health expanded insurance reimbursement for plasma-derived therapies, including IVIg and albumin for rare disease treatment.
. Policies incentivize domestic plasma collection to reduce reliance on imports and strengthen national healthcare resilience.
. Government-backed grants support R&D for innovative fractionation technologies.
. Impact: Policy support drives adoption, creates investor confidence, and ensures market sustainability.
Mergers & Acquisitions of Plasma-Derived Therapeutics
1. Grifols (U.S., 2025) acquired 14 plasma collection centers to consolidate supply networks.
2. Japanese manufacturers formed strategic alliances with global companies to enhance local production and distribution.
Buy Full Report - Access complete forecasts, regulatory updates, and M&A analysis to drive investments and corporate strategy:- https://www.datamintelligence.com/download-sample/japan-plasma-derived-therapeutics-market
Product Launches of Plasma-Derived Therapeutics
. 2025-2026 U.S.: Launch of high-potency IVIg and recombinant albumin for immunodeficiency and acute care.
. Japan: Introduction of advanced coagulation factor concentrates and next-gen immunoglobulins targeting rare hematological conditions.
Market Significance: Product innovations expand therapeutic options, improve patient outcomes, and drive hospital adoption rates.
Funding by Companies
1. Companies invested $50-120M in AI-assisted plasma fractionation and purification systems in Japan and the U.S.
2. Funding focused on capacity expansion, safety compliance, and R&D for next-gen therapies.
3. Investor Relevance: High-value capital deployment signals market confidence, growth potential, and emerging technology adoption.
Market Geographical Share
Japan: Robust domestic production (5 million liters/year) with hospital-centric demand.
U.S. & Global Influence: U.S. market approvals and supply chain developments indirectly support Japanese market expansion and collaboration opportunities.
Key Players 2026:
1. Takeda Pharmaceutical Company Limited
2. CSL Behring
3. Grifols
4. Octapharma A G
5. Kedrion Biopharma Inc
6. Baxter International
7. KM Biologics
8. Biotest AG
9. LFB Group
Conclusion
The Japan Plasma-Derived Therapeutics Market offers a compelling growth opportunity, underpinned by aging demographics, hospital demand, robust regulatory support, and strategic investments. Innovations in fractionation, M&A collaborations, and government-backed initiatives position Japan as a resilient and high-potential market for investors, executives, and healthcare stakeholders.
Related Reports
Plasma-Derived Therapeutics Market 2025
Thyroid Gland Disorders Therapeutics Market
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.